Oxygen atom1/9/2023 The table shows that substances with similar molar masses can have quite different boiling points. 1 lists the molar masses and the boiling points of some common compounds. Recall that physical properties are determined to a large extent by the type of intermolecular forces. The OH groups of alcohol molecules make hydrogen bonding possible. 2).įigure 9.2 Intermolecular Hydrogen Bonding in Methanol. 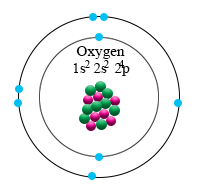
Replacing a hydrogen atom from an alkane with an OH group allows the molecules to associate through hydrogen bonding (Figure 9. This relationship is particularly apparent in small molecules and reflected in the physical and chemical properties of alcohols with low molar mass. Like the H–O–H bond in water, the R–O–H bond is bent, and the -OH portion of alcohol molecules are polar. A primary (1°) alcohol is one in which the carbon atom (in red) with the OH group is attached to one other carbon atom (in blue).Alcohols can be grouped into three classes on this basis. Some of the properties and reactivity of alcohols depend on the number of carbon atoms attached to the specific carbon atom that is attached to the -OH group. The IUPAC suffixes used in naming simple organic molecules are noted in the chart 
An example is ethanol (CH 3CH 2OH).įigure 9.1 Common Organic Functional Groups that Contain Oxygen. The IUPAC nomenclature guidelines use the suffix ‘-ol’ to denote simple compounds that contain alcohols. Because -OH is the functional group of all alcohols, we often represent alcohols by the general formula ROH, where R is an alkyl group. For example, an alcohol is an organic compound with a hydroxyl (-OH) functional group on an aliphatic carbon atom. While you will not have to formally name complete structures, you should be able to identify functional groups contained within compounds based on their IUPAC names. Figure 9.1 provides the basic organic functional groups for these compounds and the IUPAC suffix that is used to name these compounds. This includes alcohols, phenols, ethers, aldehydes, ketones, carboxylic acids, and esters. In this chapter you will be introduced to the major organic functional groups that contain oxygen. Looking at the picture, you can see there are two electrons in shell one and six in shell two.The Acidity of Carboxylic Acids Carboxylic Acids and the Formation of Salts Formation of Esters from Carboxylic Acids and Alcohols Hydrolysis of Esters 9.7 Chapter Summary 9.8 Referencesĩ.1 Introduction to Compounds that Contain Oxygen That means there are 8 electrons in an oxygen atom. for the element of OXYGEN, you already know that the atomic number tells you the number of electrons. Once one shell is full, the next electron that is added has to move to the next shell.  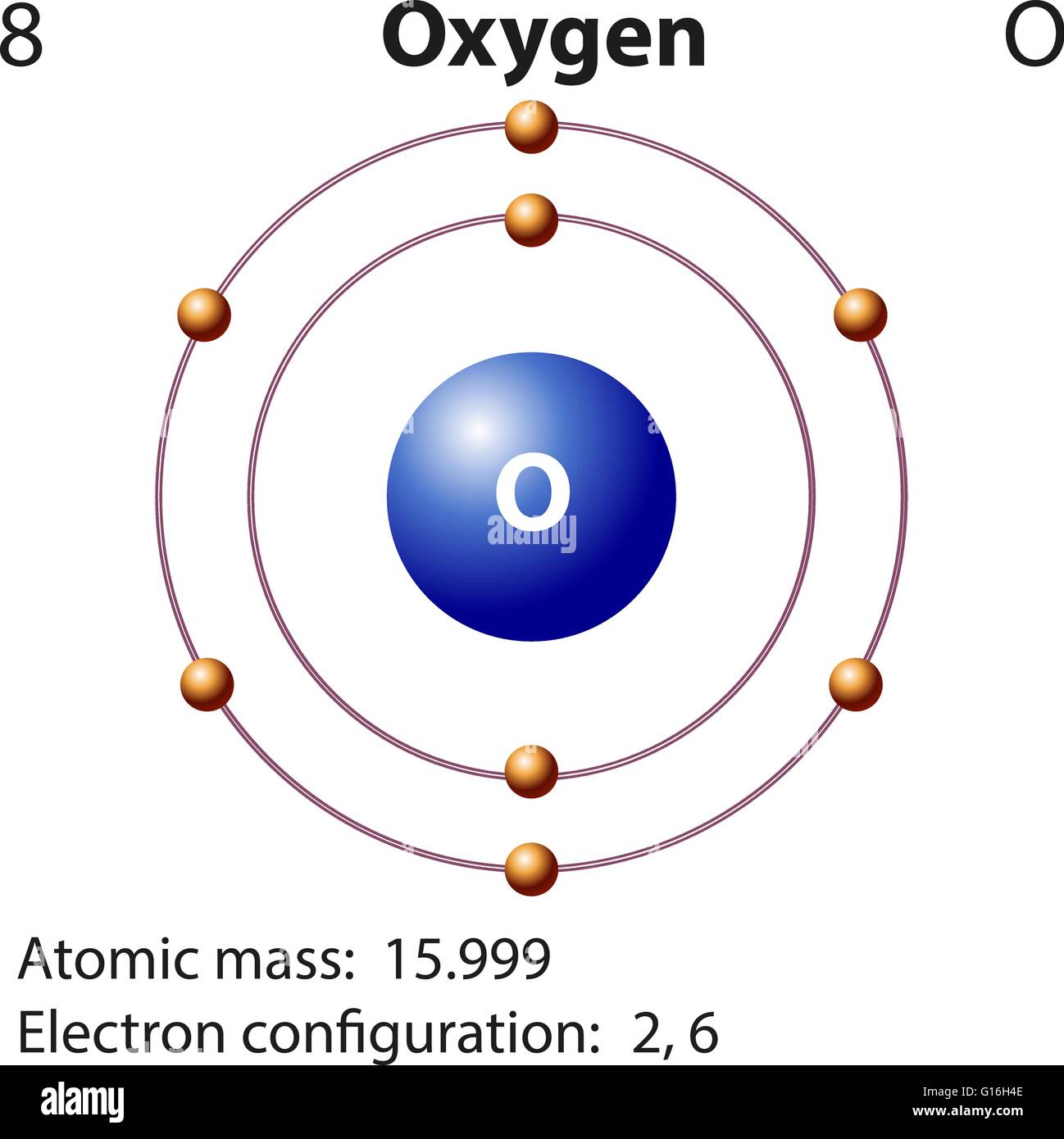
As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can hold a maximum of eight electrons. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. In the next section we're going to cover electron orbitals or electron shells. It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
